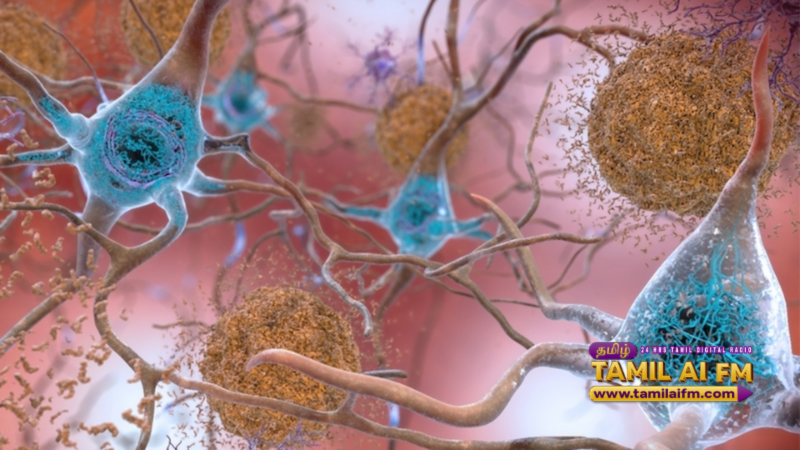
In a monumental win for medical science, the World Health Organization (WHO) and major pharmaceutical regulators have granted fast track approval to a revolutionary gene therapy treatment for Alzheimer’s disease. Clinical trials concluded earlier this week showed an unprecedented 40% reduction in cognitive decline among patients in the early stages of the disease. The treatment, developed by a joint European American research team, focuses on repairing damaged neural pathways rather than just managing symptoms.
This new therapy is being hailed as the most significant advancement in neurology in the last fifty years. Unlike previous treatments that offered marginal improvements, this gene therapy targets the underlying biological causes of dementia. Medical experts believe that with widespread implementation, the burden on global healthcare systems could be significantly reduced within a decade.
Manufacturing plants in Switzerland and Singapore have already begun scaling up production to meet expected global demand, which is estimated to be in the millions of doses. Several governments have announced plans to subsidize the treatment to ensure it remains accessible to lower income families. While the long term effects are still being monitored, today's approval marks a turning point in the global fight against neurodegenerative diseases.